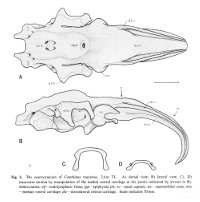
The neurocranium of Cetorhinus maximus (GUNNERUS, 1765)
by
Kunihiko Izawa and Terukazu Shibata
(published in: IZAWA, K. & SHIBATA, T. (1993): A young basking shark, Cetorhinus maximus, from Japan. Japanese Journal of Ichthyology, 40 (2): 237-245, figs 1-4)
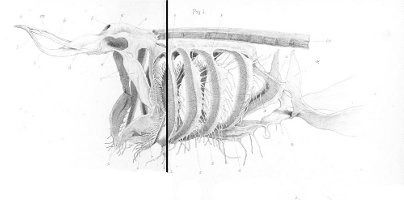
Snout and neurocranium:
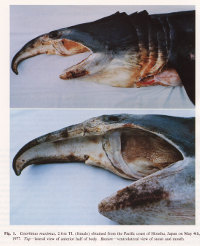
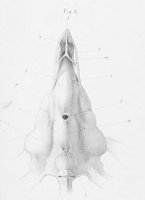
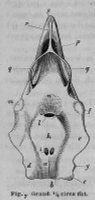
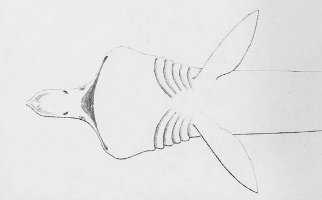
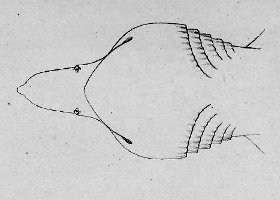
There are no previously published references to the ventral groove under the snout. A photograph of a young basking shark caught in Portobello, English Channel, in June 1950, was included in Matthews and Parker (1950, pl. 8). Apparent traces of the ventral groove can be seen in the photograph, although the authors made no mention of it. The fish length was estimated from the photograph as 2.1 -2.4 m, but is in fact believed to have been about 3 m. judging from the snout shape. The ventral groove had disappeared in individuals a little larger than the present specimen, according to the following figures; Pavesi (1874, pl. 1, fig. 3 [2.95 m]). Pavesi (1878, pl. 3, fig. 3 [3.25 m]), Carazzi (1904, fig. 1 [3.37 m]), Gervais and Gervais (1876, pl. 13, figs. 2, 3 [3,65 m]), Gudger (1935, fig. 3 [4.36 m]) and Matthews (1962, photograph of a basking shark of uncertain length). Furthermore, the ventral groove had disappeared completely from the median rostral cartilage in an 8.53 m specimen studied by Barnard (1937, fig. 1), in which the median rostral cartilage was very stout, being almost as wide as long, and had the ventral side almost flattened, although slightly depressed.
Considering the functional implications of the juvenile snout, it seems clear that newly-born basking sharks have a long snout with a ventral groove similar to the present 2.6 m specimen. Yano (1978) has speculated that its function is connected with oophagy (following observation of the same specimen dealt with here). In addition, a second function is possible. The flap-like juvenile snout with a ventral groove would assist feeding in the early, free-living young in respect to increasing the water current into the mouth and/or feeding efficiency, compensating for a presumably lesser ability to swim and feed. Photographs of a feeding basking shark in Hallacher (1977, fig. 1) show that when the fish swims with the mouth fully open, the snout receives water directly on to the ventral side, owing to the axis of the snout and head being somewhat upwardly directed. The ventral groove on the juvenile snout would clearly enhance such a feeding attitude.

Literature Cited:
Barnard, K. H. 1925. A monograph of the marine fishes of South Africa. Ann. S. Afr. Mus., 21: 1-418.
Barnard, K. H. 1937. Further notes on South African marine fishes. Ann. S. Afr. Mus., 32: 41-67, pis. 6-8.
Bigelow, H. B. and W. C. Schroeder 1948. Sharks. Pages 59-576 in A. E. Parrs and Y. H. Olsen, cds. Fishes of the western North Atlantic. Part 1, Lancelots, cyclostomcs. sharks. Sears Fundation for Marine Research, Yale Univ., Copenhagen, xvii + 576 pp.
Carazzi, D. 1904. Sulla Selache maxima Gunn. Zool. Anz., 28: 161-165.
de Buen, F. 1925. El <Cetorhinus maxinus> (Gunn.) Bol. Pescas, Madrid, 10: 237-24C, pl. 1.
de Capello, F. B. 1870. Mcmoria relativa a um exemplar de "Squalus maximus" Lin. pescado nas costas de Portugal. J. Acad. Sci. Lisbon, 2: 233-238.
Gervais, P. and H. Gervais 1876. Observations relatives a un Squale pèlerin récemment péché a Concameau. J. Zool. Paris. 5: 319-329, pls. 13-15.
Gudger, E. W. 1935. Maxillary breathing valves in the sharks Chlamydoselachus arid Cetorhinus with notes on breathing valves in thirteen marine teleosts. J. Morph., 57: 91-104.
Hallacher, L. E. 1977. On the feeding behavior of the basking shark, Ceiorhinus maximus. Env. Biol. Fish., 2: 297-298.
Kabata, Z. 1979. Parasitic Copcpoda of British fishes. The Ray Society. London. vi + 4<>8 pp.
Matthews, L. H. 1962. The shark that hibernates. New Scientist, (280): 756-759.
Matthews. L. H. and H. W. Parker 1950. Notes on the anatomy and biology of the basking shark (Cetorhinus maximus (Gunner)). Proc. Zool. Soc. Lond., 120: 535- 576. pls. 1-8.
Parker, H. W. and M. Boescman 1954. The basking shark, Cetorhinus maximus in winter. Proc. Zool. Soc. Lond., 124: 185-194, pls. 1-3.
Parker, H. W. and F. C. Stott 1965. Age, size and vertebral calcification in the basking shark, Cetorhinus maximus (Gunnerus). Zool. Mcded., 40: 305-319.
Pavesi, P. 1874. Contribusiine alia storia naturale del genere Selache. Ann. Mus. Stor. Nat. Genova, 6: 5-72, pls. 1-3.
Pavesi, P. 1878. Second contribuzione alla morfologia c systematica deiSelachi. Ann. Mus. Stor. Nat. Genova, 12: 348-418.
Pengelly, W. 1881. Basking shark in Torquay harbour. Zoologist, 5: 337-338.
Ride wood, W. G. 1921. On the calcification of the vertebral centra in sharks and rays. Philos. Trans. R. Soc. Lond., 210B: 311-407.
Springer, S. and P. W. Gilbert 1976. The basking shark. Cetorhinus maximus, from Florida and California, with comments on its biology and systematic. Copeia, 1976: 47-53.
Yano, K. 1976. Same no sekai (The world of sharks). Shinchosha, Tokyo. 230 pp. (In Japanese.)
Yano, K. 1978. Ubazame ni tsuite (On basking shark). Bansairui Kenkyu Renrakukai, no. 3. (In Japanese.)
---------------------------------------------------------------------------------------------------------------------------------
List of References from shark-references.com